The His tag 248 is by far the most popular affinity tag for purification of recombinant proteins. SEC resins used for purification of biomolecules have a designed fractionation range which defines the range of molecular weights M r that have partial access to the pores of the particle and can be separatedFractionation ranges of M r from 100 to 7000 are suited to separation of peptides and other small biomolecules while a resin with a fractionation range of 100 000 to 300 000 is.
Protein Affinity Chromatography Caframo Lab Solutions
Recombinant Purification Definition Biologicscorp
Facebook
Hober S Nord K and Linhult M.
Column chromatography protein purification. This will elute all bound proteins from the column. Purification of the recombinant protein using spin column chromatography with Norgens proprietary resin as an ion-exchanger Each spin column is able to purify up to 12 mg of recombinant proteins from 100 mL of culture. It actively plays a role in isolating major constituents and helps disintegrates complex mixtures.
Broad product selectionprepared affinity and ion exchange supports for the purification and enrichment of proteins and antibodies as well as activated supports for custom ligand immobilization. Flash column chromatography has firmly established itself as an effective and reliable purification method allowing scientists across several disciplines to extract compounds from mixtures for. General sequence of protein purification procedures Preparation of equipment and reagents For protein purification you need the equipment and reagents listed below.
Compounds move through the column at different rates allowing them to be separated into fractions. Kinsland in Comprehensive Natural Products II 2010 919621 Immobilized metal affinity chromatography. Thus the workflow below is given as a generalized IEX workflow and particular running conditions for anion exchange chromatography may be adjusted to best suit your protein of interest the buffer system and the anion exchange resin chosen.
Column chromatography in chemistry is a chromatography method used to isolate a single chemical compound from a mixture. Chromatography column chromatography protein purification Chromatography is based on the principle where molecules in mixture applied onto the surface or into the solid and fluid stationary phase stable phase is separating from each other while moving with the aid of a mobile phase. All ion exchange chromatography relies on electrostatic interactions between the resin functional groups and proteins of interest.
At Cytiva we always measure the dynamic binding capacity to show what you can really expect from the column. Ni 2 is most commonly used for his-tag purification since it gives a high yield. Most of these methods do not require the denaturing of proteins.
The MAbPac Protein A column has been successfully tested through 2000 cycles without loss of binding capacity. The kit is designed to purify both acidic and basic proteins. Chromatography is an important biophysical technique that enables the separation identification and purification of the components of a mixture for qualitative and quantitative analysis.
The MagneGST Protein Purification System Technical Manual includes several protocols including small-scale purification using the MagneGST Cell Lysis Reagent 1ml cultures small-scale protein purification from cells lysed by sonication or other methods and large-scale purification 150ml cultures or an equivalent amount of cleared lysate. Proteins vary hugely in their properties and the different types of column chromatography allow you to exploit those differences. DBC takes into account the risk of protein losses during purification with a column.
Protein purification is vital for the specification of the function structure and interactions of the protein of interest. Column chromatography is one of the most common methods of protein purification. His-tagged protein purification requires the His-tag and Ni-NTA interaction which is based on the selectivity and high affinity of Ni-NTA nickel nitrilotriacetic acid resin for proteins containing an affinity tag of eg.
More formatsmagnetic beads 96-well filter plates loose resins spin columns and kits and FPLC cartridges to facilitate screening and other small-scale experiments as well as process-scale. However for protein purification the stability of the protein is the most important consideration in choosing purification conditions and thus the most appropriate column for protein binding. A high dynamic binding capacity yields in more purified proteinmL resin in a prepacked column and with that reduces the costmg protein.
Column Chromatography A Versatile Approach. NTA which has four chelating sites for nickel ions binds nickel more tightly than metal-chelating purification systems like IDA which. Flexible and intuitive column chromatography system to meet your purification challenges in research applications.
In general affinity chromatography can be effectively used to isolate a protein that recognizes a certain group by 1 covalently attaching this group or a derivative of it to a column 2 adding a mixture of proteins to this column which is then washed with buffer to remove unbound proteins and 3 eluting the desired protein by adding a high concentration of a soluble form of the. 1 2 A widely employed method utilizes immobilized metal-affinity chromatography IMAC to purify recombinant proteins containing a short affinity tag consisting of polyhistidine residues. Using Co 2 can give higher purity but with a lower yield.
4 2 6 0 Protein purification Purification of a His6-tagged Green Fluorescent Protein GFP. A powerful purification method involves the use of peptide affinity tags which are fused to the protein of interest and used to expedite protein purification via affinity chromatography. Typically the tag is composed of 610 consecutive histidines at either terminus of the protein of interest often separated by a protease-cleavage site.
At 2 mLmin a complete titer analysis takes 2 min. The Russian botanist Mikhail Tswett coined the term chromatography in 1906. We cannot deny how column chromatography is the best way to purify and separate different phases of matter.
PhyNexus provides life science researchers automation solutions to high throughput sample processing in drug discovery and screening using our PhyTip columns with Dual Flow Chromatography technology. Therefore it is necessary to determine whether conditions required for binding to either form of ion exchange chromatography affect protein stability and function. The MAbPac Protein A column has a fast cycle time.
Protein purification is a series of processes intended to isolate one or a few proteins from a complex mixture usually cells tissues or whole organisms. Automated Protein Plasmid and AAV Purification with up to 95 Yields. Like many of the techniques on this site it is as much an art form as a science.
Bio-Rad has his-tag resins and his-tag purification kits that are precharged with Ni 2 for fast. Beads in the chromatography column are cross-linked to ligands that bind specifically to the target protein. Sonicator or French Press Centrifuge medium speed 30-70Kg eg.
ÄKTA pure 25 and ÄKTA pure 150 protein purification systems allows not only multistep fast and efficient protein purification but also unattended operations by efficiently automating your purification tasks. Chromatography is able to separate substances based on differential adsorption of compounds to the adsorbent. Six consecutive Histidine residues.
XAfter all specifically bound protein has been eluted from the column wash the column with 10 bed volumes 6 ml elution buffer 3. Protein A Chromatography for Antibody Purification. Chromatography Column chromatography is one of the most common methods of protein purification.
Moreover it also assists in the estimation of drugs in drugs formulation etc. His-tagged protein is then eluted with a higher concentration of imidazole. Affinity chromatography is a very useful technique for polishing or completing the protein purification process.
J-20 or Avanti J-25 Appropriate centrifugation tubes Chromatographic system comprising of as a minimum pump and fraction.
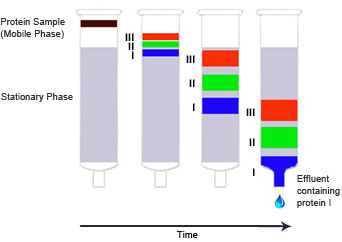
Introduction To Chromatography Bio Rad Laboratories

Affinity Chromatography Which Tag To Use Cube Biotech

How Column Chromatography Works To Separate Proteins Goldbio

Affinity Chromatography Wikipedia

Protein Purification Workflow The Bumbling Biochemist

Protein Purification And Analysis Solubility Of Proteins Important
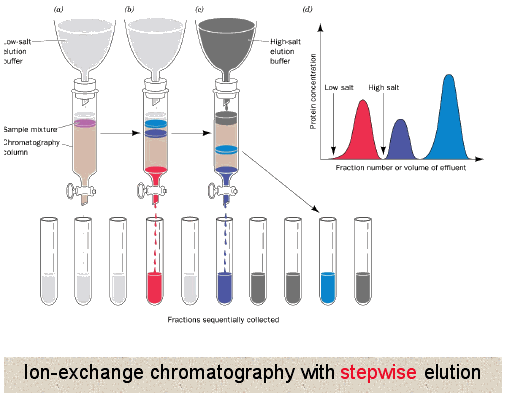
Column Chromatography

Column Chromatography Biotechnology Foundations 2nd Edition Openstax Cnx



